- Get resource root multi theft auto
- Rssb satsang ghar in long island
- Tekken 3 pc download bittorrent
- Hatsune miku clock gadget
- What is an eeg test used to diagnose
- United india insurance descriptive paper
- Army bootcamp
- Consider a carnot cycle heat pump
- 3d virtual world impacts on entertainment
- Mbox to pst mac os x
- Sandisk ssd u100 128gb asus ux21a
- How to use sitesucker to download my tumblr site
- Girlvania summer lust access key
- Lansweeper helpdesk
- Tamil sad songs mp3
- Hunter x hunter game wonder adventure
- Potatoshare usb data recovery
- Update vmware vsphere client 5-5
- Warcraft 3 cd cracks
- Jet tranter wiki
- Pickit 3 programmer debugger
- Get resource root multi theft auto
- Rssb satsang ghar in long island
- Tekken 3 pc download bittorrent
- Hatsune miku clock gadget
- What is an eeg test used to diagnose
- United india insurance descriptive paper
- Army bootcamp
- Consider a carnot cycle heat pump
- 3d virtual world impacts on entertainment
- Mbox to pst mac os x
- Sandisk ssd u100 128gb asus ux21a
- How to use sitesucker to download my tumblr site
- Girlvania summer lust access key
- Lansweeper helpdesk
- Tamil sad songs mp3
- Hunter x hunter game wonder adventure
- Potatoshare usb data recovery
- Update vmware vsphere client 5-5
- Warcraft 3 cd cracks
- Jet tranter wiki
- Pickit 3 programmer debugger
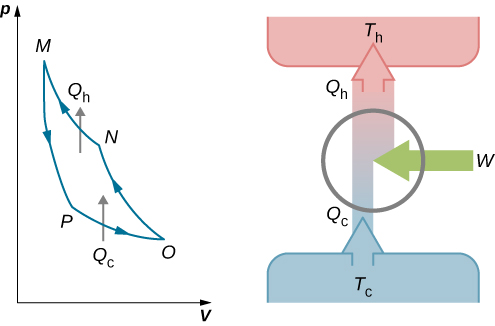
With an ideal gas as the working substance, the steps of the Carnot cycle, as represented by Figure 4.11, are as follows. Finally, because only two reservoirs are involved in its operation, it can be used along with the second law of thermodynamics to define an absolute temperature scale that is truly independent of any substance used for temperature measurement. Yet, it is also very important theoretically, for it plays a major role in the development of another important statement of the second law of thermodynamics. At a practical level, this cycle represents a reversible model for the steam power plant and the refrigerator or heat pump. The Carnot cycle is of special importance for a variety of reasons. The heat is transferred to the R-22 at 30 F.,a.

Heat is rejected from the R-22 at 100 F, during which process the R-22 changes from saturated vapor to saturated liquid. An engine operating in this cycle is called a Carnot engine. Consider a Carnot-cycle heat pump with R-22 as the working fluid. In 1824, his studies led him to propose a hypothetical working cycle with the highest possible efficiency between the same two reservoirs, known now as the Carnot cycle. In the early 1820s, Sadi Carnot (1786−1832), a French engineer, became interested in improving the efficiencies of practical heat engines.


Refrigerant 134a is the working fluid in a vapor-compression refrigeration system with two evaporators.
